
Effect of the non-conserved N-terminus on the DNA binding activity of the yeast TATA binding protein. |
Department of Molecular Genetics and Biochemistry
University of Pittsburgh School of Medicine
Pittsburgh, PA 15261
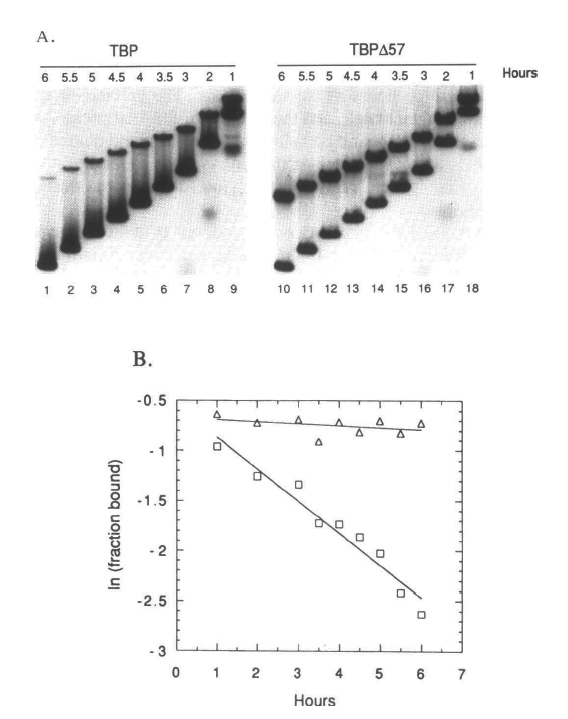
| We have studied the DNA binding activity of recombinant yeast TATA Binding Protein (TBP) with particular interest in the role played by the nonconserved N-terminal domain. By comparing the DNA binding activity of wild type yeast TBP with a mutant form of TBP that lacks the non-conserved N-terminal domain (TBPd57), we have determined that the N-terminus of TBP alters both the shape and the stability of the TBP-DNA complex. Measurements of the DNA bending angle indicate that the N-terminus enhances the bending of the DNA that is induced by TBP binding and greatly destabilizes the TBP-DNA complex during native gel electrophoresis. In solution, the N-terminus has only a slight effect on the equilibrium dissociation constant and the dissociation rate constant. However, the N-terminal domain reduces the association rate constant in a temperature dependent manner and increases the apparent activation energy of the TBP-DNA complex formation by 3 kcal/mole. These data suggest that a conformational change involving the N-terminus of TBP may be one of the isomerization steps in the formation of a stable TBP-DNA complex. |
