Department of Molecular Genetics and Biochemistry
University of Pittsburgh School of Medicine
Pittsburgh, Pennsylvania 15261
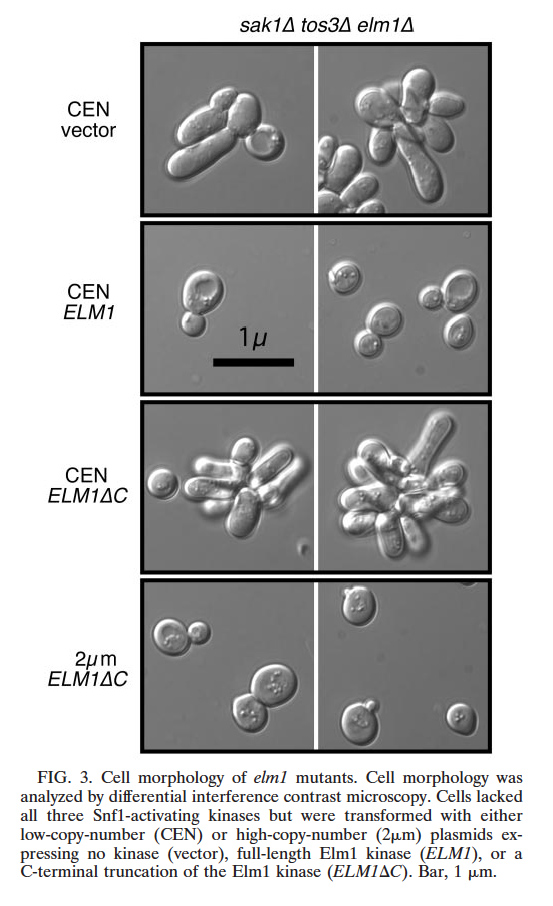
| In Saccharomyces cerevisiae, the Snf1 kinase can be activated by any one of three upstream kinases, Sak1, Tos3 or Elm1. All three Snf1-activating kinases contain serine/threonine kinase domains near their N-termini and large C-terminal domains with little sequence conservation and previously unknown function. Deletion of the C-terminal domains of Sak1 and Tos3 greatly reduces their ability to activate the Snf1 pathway. In contrast, deletion of the Elm1 C-terminal domain has no effect on Snf1 signaling but abrogates the ability of Elm1 to participate in the morphogenetic checkpoint signaling pathway. Thus, the C-terminal domains of Sak1, Tos3 and Elm1 help to determine pathway specificity. Additional deletion mutants of the Sak1 kinase revealed that the N-terminus of the protein is essential for Snf1 signaling. Deletion of 43 amino acids from within the N-terminus of Sak1 (residues 87-129) completely blocks Snf1 signaling and activation loop phosphorylation in vivo. The Sak1 kinase domain (lacking both N-terminal and C-terminal domains) is catalytically active and specific in vitro but is unable to promote Snf1 signaling in vivo. Our studies indicate that the kinase domains of the Snf1-activating kinases are not sufficient by themselves for their proper function and that the non-conserved N-terminal and C-terminal domains are critical for the biological activities of these kinases. |