Protein Biophysics
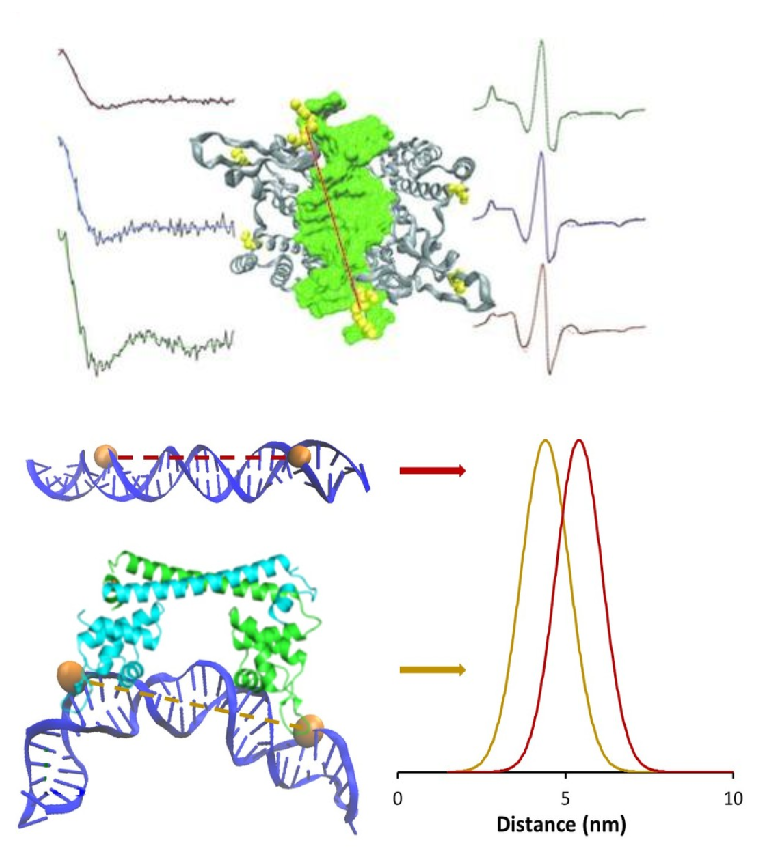
Protein-DNA Interactions
The interactions between proteins and specific sequences of DNA lie at the heart of many cellular processes such as DNA repair, transcription of new proteins, and cellular defense.
EcoRI is a restriction endonuclease that binds to and cleaves viral DNA at a specific six base pair sequence. While this lethal cleavage activity protects the cell from viral infection, an amazing binding specificity is required so that the cell’s own DNA is not cleaved. We have revealed the structural and dynamical details that underlie the binding and cleavage specificity by comparative distance and measurements on the protein bound to specific and non-cognate sequences of DNA. Such information informs us about the molecular origins of the specificity of protein-DNA interactions. In addition, we have discovered a new metal-binding site in EcoRI and delineated the mechanistic role of metal binding at this site in the modulation of DNA cleavage.
Understanding the atomic level details of protein-DNA interactions is also important in order to develop new antibiotics, which can be used to combat with drug-resistant bacteria. Copper is an essential nutrient for microbes. However, excess copper causes cell toxicity and cell death. Due to the toxicity of copper, microbes have developed tightly regulated mechanisms to maintain the copper levels inside cells. CueR is a metal sensor protein present in the microbes which senses copper with zeptomolar affinity. In the presences of copper, CueR binds to DNA to initiate the generation of proteins that remove excess copper ions from the cell. We are currently applying site directed Cu(II) spin labeling methods to probe the CueR-DNA interactions, both in-vitro and in cells. By understanding the conformational changes accompanying such protein-DNA complexes, new inhibitors can be designed which can potentially modulate the DNA interaction, leading to loss of the protective mechanism.
Collaborators: Prof. Sharon Ruthstein (Bar Ilan University, Israel), Prof. Linda-Jen Jacobson (Pitt)
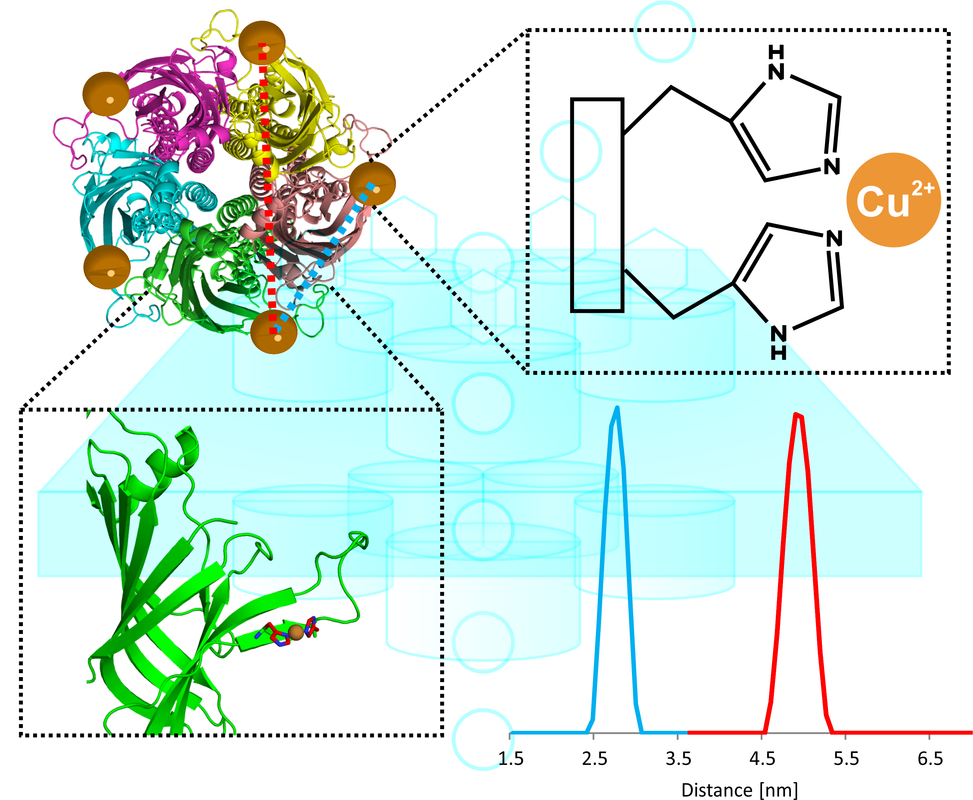
Pentameric Ligand Gated Ion Channel
Electron spin resonance distance measurements along with site-directed spin labeling offers the potential to study complex biological systems, such as ligand gated ion channels (LGICs). These channels regulate the flow of ions into cells upon the binding of an agonist, which induces the channel to open or close. LGICs typically have three distinct conformational states; A resting state in which the channel is closed, an open state in which agonist binding causes the channel to open and ions to flow through, and a desensitized state in which the channel is non-conducting after prolonged exposure to the agonist. Our group aims to cultivate a more thorough understanding of the specific conformational changes that occur as the ion channel transitions between each state.
Our group has investigated systems such as GlyR, a glycine receptor that permits the flux of chloride ions across cell membranes, by examining Cu(II)’s inhibiting effect on channel activity. Recently, we have turned our attention to the prokaryotic Erwinia chrysanthemi ion channel, ELIC. We combined MTSSL-based ESR and NMR constraints to provide insight into the desensitization mechanism.
However, such ESR experiments are limited by the inherent flexibility of the typical nitroxide spin probes that were used. Consequently, the measured distances and dynamics are difficult to relate to the actual protein backbone and a need exists for more precise methods of measurement. We aim to solve this problem by applying a double histidine (dHis) motif. Using the dHis Motif we can measure distance with unprecedented precision. Distance measurements in the presence and absence of ELIC’s agonist, propylamine, can shed new light on the structural and conformational changes that occur during the ion channel’s opening and closing and help us understand the gating mechanism that governs this important biological function.
Collaborator: Pei Tang (Pitt)
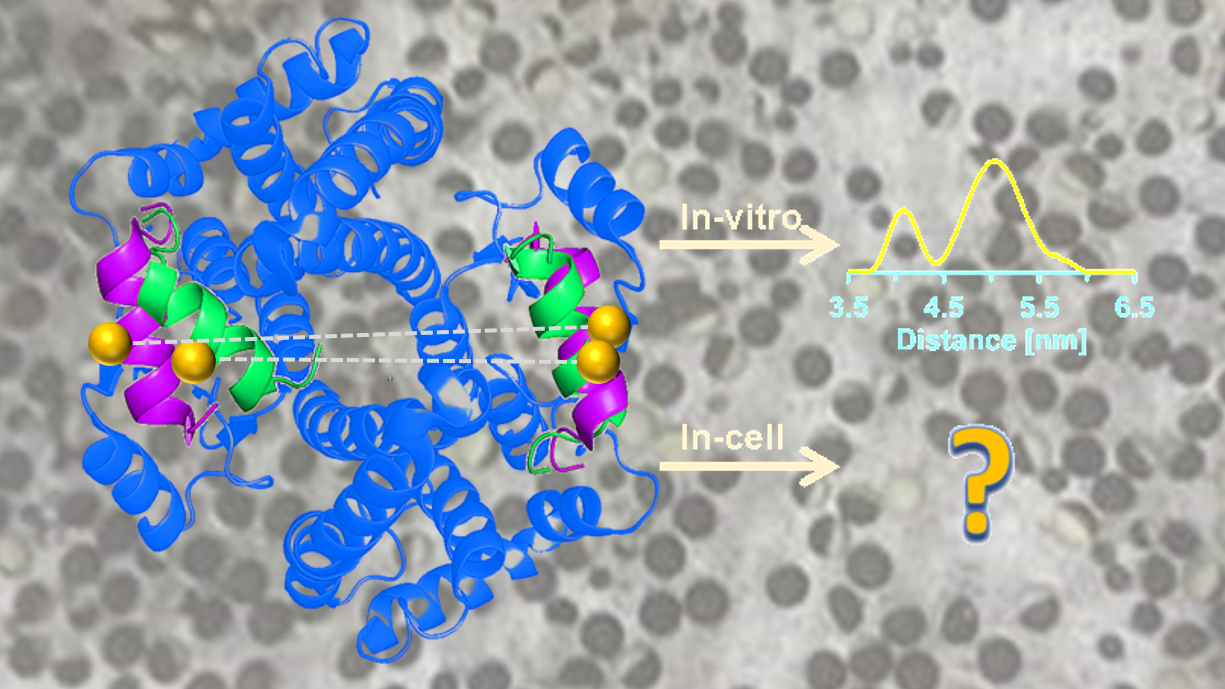
In-Cell Protein Dynamics
Our broad goal in this area is to develop simple and broadly applicable strategies to enable measurements of protein structure, interactions, and dynamics in-cell by ESR. Distance measurements in-cell by pulsed ESR present tremendous opportunities to study proteins in a more native environment that is irreproducible in vitro. Inside the cell, it is estimated that the concentration of biological macromolecules is in the range of 80-400 mg/mL. This “crowded” environment can affect not only the structure and dynamics of biological macromolecules, but also various biological equilibria such as protein folding, aggregation, binding of small molecules, enzymatic activity and protein-protein interactions.
The in-cell environment is harsh towards the typical nitroxide radical used in DEER experiments. We have developed simple procedures to enhance the lifetime of the nitroxide radical in cell by using an oxidizing agent. Using this procedure, we can perform distance measurements on doubly nitroxide labeled proteins, at different incubation times in the cellular environment. With this success, we will elucidate the structure and dynamics of the detoxification enzyme glutathione s-transferase (GST), which plays an important role in cellular defence. GSTs catalyze the conjugation of glutathione to various endogenous and exogenous electrophiles. This results in a deactivation of the reactive centers of such electrophiles, providing protection from a wide range of xenobiotics. We have recently combined ESR and NMR distance constraints with molecular modelling to resolve the structure of the ligand free conformations of GST in vitro. When the ligand S-hexyl glutathione was introduced, there was a clear shift in the distance distribution favouring one dominant conformation. Room temperature ESR indicates the second conformation has high mobility potentially enabling the enzyme’s high degree of substrate promiscuity.
However, it is currently unknown whether the same dynamic processes are present in the cell. In addition to serving as important detoxification enzymes, the enzymes also interact with cellular signalling pathways that regulate apoptosis, consequently they have been implicated in modulating apoptosis of cancer cells. Although this interaction has been characterized in vitro, there have not been any studies that directly detect this important interaction in vivo. Our current research focuses on bridging this gap in knowledge.
Collaborator: Gordon Rule (CMU)
