Ionic liquids • Zhe Ren , Tom Brinzer , Jordan Kelly , Emma Coate
Ionic liquids are a promising platform for selective, high capacity, low energy-cost gas separation technologies. Progress in their development, however, is hindered by a lack of fundamental understanding of the structure and dynamics of these complex fluids as well as how they solvate molecular adsorbates. The rich variety of intermolecular interactions between the cations and anions cause unusual mesoscopic and macroscopic behaviors in these fluids. Ultrafast vibrational spectroscopy has the ability to interrogate molecular structure and picosecond intermolecular motions underlying these larger time- and length-scale phenomena. 2D-IR experiments will investigate both the bonding between cation and anion as well as the picosecond dynamics of CO2 in imidazolium ionic liquids.
|
Ion sensing in EF-Hand peptides • Samrat Dutta , Sunayana MitraIn all forms of life, proteins chelate metal ions to perform myriad functions. The ions may play a structural role (e.g. zinc finger proteins), a catalytic role (e.g. Fe in cytochromes), or a dynamic role (e.g. passing K+ ions through a membrane). Many cellular signaling processes are mediated or transmitted by the Ca2+ ion. The signal can be carried directly by Ca2+ after ion channel opening or indirectly as a secondary messenger, for example in G-protein coupled receptors. Almost all of the Ca2+ modulated events involve the binding of Ca2+ to a domain called the EF-hand. EF-hand is utilized by prokaryotes as well as eukaryotes (plants, animals, and fungi), and more than 1000 EF-hand Ca2+ binding proteins are known. Xray crystallography has provided high resolution structures of several important Ca2+ binding proteins, but, during function, the protein structures are dynamic. Elucidation of these stuctural changes will reveal how these proteins sense changes in ion concentration and how local binding events are transmitted to the global reorganization of the protein.
In large part, this lack of understanding is due to a lack of experimental tools with the appropriate temporal- and spatial-resolution. Ultrafast vibrational spectroscopy addresses exactly these needs. Its time-resolution is sub-picosecond and can give residue specific information. The overarching goal of this project is to develop ultrafast vibrational spectroscopy as a general tool to interrogate the inner-sphere coordination of polypeptides with biologically important metal cations. |
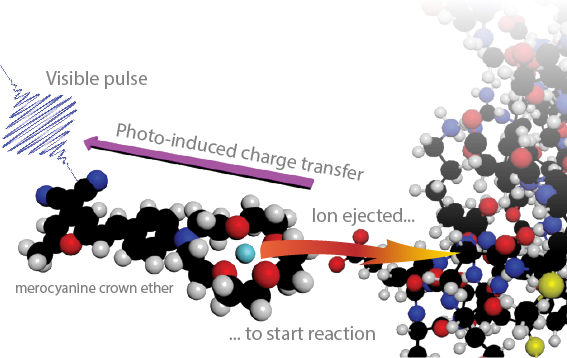
Ion-Concentration Jump Experiment • Samrat DuttaIon uptake in EF hand is too fast for stopped flow experiments and single-molecule spectroscopy to time-resolve.
None of the standard time-domain techniques can break into the sub-microsecond regime, which is important to resolve elementary kinetic steps of the Ca2+ induced conformational change. A "caged-calcium" compound will be developed to create a photoinduced change in the ion concentration on a sub-picosecond timescale. This ion-concentration jump experiment will probe the reorganization of synthetic EF-hand polypeptides over the full range of timescales (picoseconds to microseconds) with residue specificity. We will learn about the elementary kinetic steps of ion binding, reorganization of the binding pocket, and the transmission of the binding signal to the global conformation of the domain. |
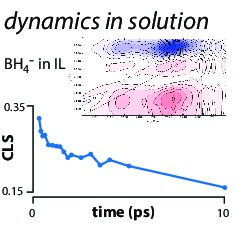
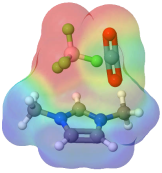
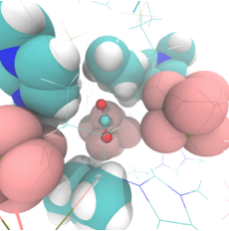
Hydride transfers from BH4- • Clinton Johnson, Molly WagnerBH4- is a strong chemical reductant. Using ultrafast vibrational spectroscopy, we are probing how solvation controls the chemistry. We are interested in establishing both the dynamics at equilibrium (using 2D-IR spectroscopy) and the kinetics of reaction after photoexcitation.
|
Collaborations
Electronic structure of CO2 in ionic liquidsIn collaboration with Prof. Daniel Lambrecht's group (Pitt, Chemistry), we are developing electronic structure methods to gain insight into the physical origin of the vibrational frequency shifts that CO2 reports. A decomposition scheme based on Absolutely Localized Molecular Orbitals (ALMOs) allows us to separate the many contributing effects – geometrical distortion, polarization, electrostatics, and charge transfer.
|
Molecular dynamics of CO2 in ionic liquidsIn collaboration with Prof. Steve Corcelli's group (Notre Dame, Chemistry), we are using molecular dynamics simulations to assign and interpret the molecular motions that are reported in the 2D-IR spectroscopy. Insight into the molecular interactions between CO2 and ionic liquids is important for the design of future carbon capture systems.
|
CO2 reduction in green solventsIn collaboration with Prof. John Keith (Pitt, Chem. Eng.), we are investigating the role of solvents in controlling hydride transfers from strong chemical reductants like BH4-. The insight from this project is important for converting CO2 into a renewable fuel stock.
|
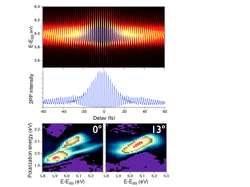
Multidimensional photoelectron spectroscopy In collaboration with Prof. Hrvoje Petek's group (Pitt, Physics), we are developing multidimensional photoelectron spectroscopies.
|